
FARADAY'S LAW. NaCl (s) → Na + (l) + Cl – (l) E° R = V E° O = V 2 Cl - (l) → Cl 2(g) + 2e - 2 Na + (l) + 2e - → 2 Na (s) Electrolytic cell. - ppt download

1 Suppose we had a 0.3A current producing 15mL of hydrogen gas in 6 min and 30 seconds. How much charge did we have per one mole of electrons? We need. - ppt download

1 mol of charge = 1 Faraday = 96500 coulombs. Represent the cell and the standard emf of the cell having the following reaction. 2Cr(s) +3Ca(aq) = 2Cr + (aq) + 3Cd()

The mass of the substance deposited by 1 faraday of electricity is equal to 11 grams. The value of electrochemical equivalent is: A. 11 B. 11 x 96500 - Correct Answers 11 96500 D. data insufficient

SOLVED:The faraday is a unit of charge frequently encountered in electrochemical applications and named for the British physicist and chemist Michael Faraday. It consists of 1 mole of elementary charges. Calculate the

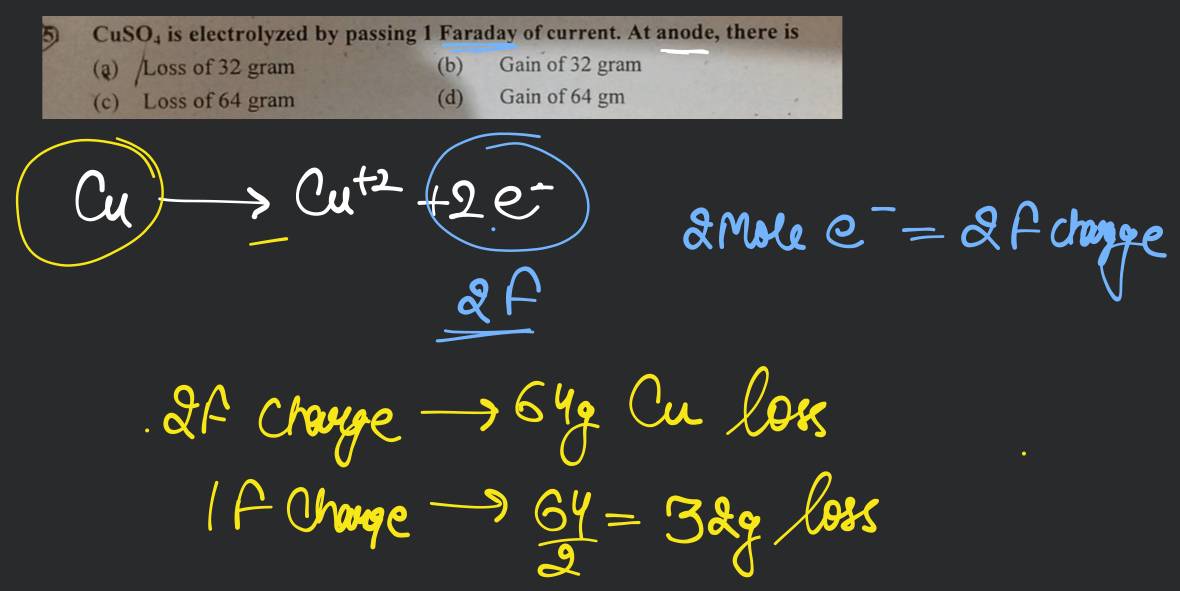















.png)